Research Area B - Publications 2011
20-Dec-2011
PNAS, online article

The molecular chaperone αB-crystallin, the major player in maintaining the transparency of the eye lens, prevents stress-damaged and aging lens proteins from aggregation. In nonlenticular cells, it is involved in various neurological diseases, diabetes, and cancer. Given its structural plasticity and dynamics, structure analysis of αB-crystallin presented ...
15-Dec-2011
Plant Signaling & Behavior, online article

Import of nuclear encoded proteins into chloroplast is an essential and well-regulated mechanism. The cytosolic kinases STY8, STY17 and STY46 have been shown to phosphorylate chloroplast preprotein transit peptides advantaging the binding of a 14-3-3 dimer. Analyses of sty8 sty17 sty46 mutant plants revealed a role for the kinases in chloroplast differentiation, ...
09-Dec-2011
Journal of Experimental Botany, online article

Previously, the OEP16.1 channel pore in the outer envelope membrane of mature pea (Pisum sativum) chloroplasts in vitro has been characterized to be selective for amino acids. Isolation of OEP16.2, a second OEP16 isoform from pea, in the current study allowed membrane localization and gene expression of OEP16 to be followed throughout seed development and ...
05-Dec-2011
PNAS, online article
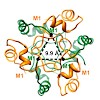
The molecular chaperone αB-crystallin, the major player in maintaining the transparency of the eye lens, prevents stress-damaged and aging lens proteins from aggregation. In nonlenticular cells, it is involved in various neurological diseases, diabetes, and cancer. Given its structural plasticity and dynamics, structure analysis of αB-crystallin presented ...
25-Nov-2011
Plant Molecular Biology, online article
Calmodulin (CaM) is a ubiquitous sensor/ transducer of calcium signals in eukaryotic organisms. While CaM mediated calcium regulation of cytosolic processes is well established, there is growing evidence for the inclusion of organelles such as chloroplasts, mitochondria and peroxisomes into the calcium/calmodulin regulation network. A number of CaM-binding ...
15-Nov-2011
Mol. Biol. Cell, online article
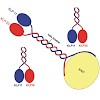
The heterotrimeric structure of kinesin-2 makes it a unique member of the kinesin superfamily; however, molecular details of the oligomer formation are largely unknown. Here we demonstrate that heterodimerization of the two distinct motor domains KLP11 and KLP20 of Caenorhabditis elegans kinesin-2 requires a dimerization seed of merely two heptads at the C ...
11-Nov-2011

Nature provides a rich source of bioactive compounds comprising a diverse set of electrophilic core structures that are poised to react with corresponding nucleophilic residues such as serine and cysteine in enzyme active sites.These residues are usually relevant for catalysis and therefore display fine-tuned reactivity towards their dedicated substrates.We and ...
02-Nov-2011
FEBS Letters, online article

Chloroplasts and mitochondria are central to crucial cellular processes in plants and contribute to a whole range of metabolic pathways. The use of calcium ions as a secondary messenger in and around organelles is increasingly appreciated as an important mediator of plant cell signaling, enabling plants to develop or to acclimatize to changing environmental ...
04-Sep-2011
Nature Methods, online article

Maintenance of cellular protein homeostasis (proteostasis) depends on a complex network of molecular chaperones, proteases and other regulatory factors. Proteostasis deficiency develops during normal aging and predisposes individuals for many diseases, including neurodegenerative disorders. Here we describe sensor proteins for the comparative measurement of ...
04-Sep-2011
Nature Structural & Molecular Biology, online article
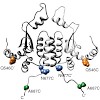
In eukaryotes, the essential dimeric molecular chaperone Hsp90 is required for the activation and maturation of specific substrates such as steroid hormone receptors, tyrosine kinases and transcription factors. Hsp90 is involved in the establishment of cancer and has become an attractive target for drug design. Here we present a structural characterization of the ...
28-Jul-2011
Plant Physiology, online article

In Arabidopsis (Arabidopsis thaliana), transit peptides for chloroplast-destined preproteins can be phosphorylated by the protein kinases STY8, STY17, and STY46. In this study, we have investigated the in vitro properties of these plant-specific kinases. Characterization of the mechanistic functioning of STY8 led to the identification of an essential threonine in ...
21-Jun-2011
ChemBioChem, online article
Due to their endosymbiotic origin, chloroplasts are completely reliant on post-translational import of their resident proteins.1 Integral membrane proteins comprise approximately 20 % of the total estimated chloroplast proteome.2, 3 Based on several proteomic analyses, 20 % of all membrane proteins are located in the inner envelope (IE).4–6 Judging from the ...
18-May-2011
Molecular Plant, online article

Most chloroplast and mitochondrial proteins are synthesized in the cytosol of the plant cell and have to be imported into the organelles post-translationally. Molecular chaperones play an important role in preventing protein aggregation of freshly translated preproteins and assist in maintaining the preproteins in an import competent state. Preproteins can ...
01-May-2011
Journal of Endocytobiosis and Cell Research, online article

A major problem for chloroplast research remains that plant species which are utilized in order to obtain a high amount of isolated intact chloroplasts are not sequenced on a genomic base. Thus it is difficult to combine the data from biochemical experiments with genomic data and employ techniques such as MS to identify low abundant proteins. There are plant ...
Total Chemical Synthesis of an Integral Membrane Enzyme: Diacylglycerol Kinase from Escherichia coli
18-Apr-2011
Angew. Chemie, online article

Recent progress in chemical protein synthesis has provided access to many small to medium-sized proteins. However, the highly important class of membrane proteins comprising multimembrane-spanning receptors and ion channels as well as integral membrane enzymes remains elusive with regard to chemical synthesis. Only certain moderately sized membrane proteins have ...
13-Apr-2011
The molecular chaperone Hsp90 regulates the activity and stability of a set of client proteins. Despite prog- ress in understanding its mechanism, the interaction of Hsp90 with clients has remained enigmatic. Now, in a recent issue of Molecular Cell, Street and coworkers present results that integrate the client in the Hsp90 chaperone cycle.
16-Feb-2011
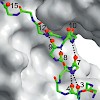
Understanding the mechanism of protein folding requires a detailed knowledge of the structural properties of the barriers separating unfolded from native conformations. The S-peptide from ribonuclease S forms its Alpha-helical structure only upon binding to the folded S-protein. We characterized the transition state for this binding-induced folding reaction at ...
24-Jan-2011
Protein Science, online article

Protein disulfide isomerase (PDI) supports proinsulin folding as chaperone and isomerase. Here, we focus on how the two PDI functions influence individual steps in the complex folding process of proinsulin. We generated a PDI mutant (PDI‐aba′c) where the b′ domain was partially deleted, thus abolishing peptide binding but maintaining a PDI‐like redox potential. ...
19-Jan-2011

Human epithelia are permanently challenged by bacteria and fungi, including commensal and pathogenic microbiota. In the gut, the fraction of strict anaerobes increases from proximal to distal, reaching 99% of bacterial species in the colon. At colonic mucosa, oxygen partial pressure is below 25% of airborne oxygen content, moreover microbial metabolism causes ...
19-Jan-2011
Nature, online article

Human epithelia are permanently challenged by bacteria and fungi, including commensal and pathogenic microbiota. In the gut, the fraction of strict anaerobes increases from proximal to distal, reaching 99% of bacterial species in the colon. At colonic mucosa, oxygen partial pressure is below 25% of airborne oxygen content, moreover microbial metabolism causes ...
09-Jan-2011
Nature Strucrural & Molecular Biology, online article
The endoplasmic reticulum is the site of folding, assembly and quality control for proteins of the secretory pathway. The ATP-regulated Hsp70 chaperone BiP (heavy chain–binding protein), together with cochaperones, has important roles in all of these processes. The functional cycle of Hsp70s is determined by conformational transitions that are required for ...










