Cdc14 phosphatase promotes segregation of telomeres through repression of RNA polymerase II transcription
21-Oct-2011
Nature Cell Biology, 2011, doi:10.1038/ncb2365, published on 21.10.2011
Nature Cell Biology, online article
Nature Cell Biology, online article
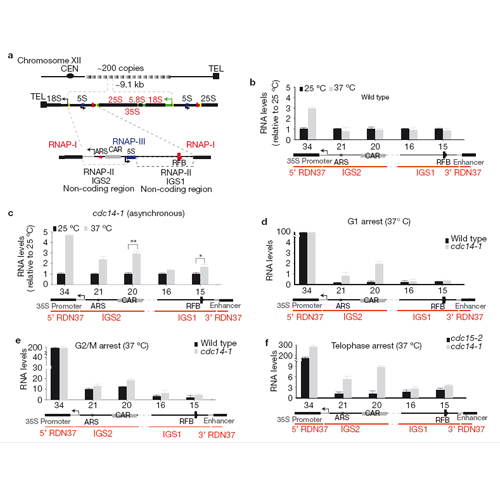
Kinases and phosphatases regulate messenger RNA synthesis through post-translational modification of the carboxy-terminal domain (CTD) of the largest subunit of RNA polymerase II (ref. 1). In yeast, the phosphatase Cdc14 is required for mitotic exit2,3 and for segregation of repetitive regions4. Cdc14 is also a subunit of the silencing complex RENT (refs 5,6), but no roles in transcriptional repression have been described. Here we report that inactivation of Cdc14 causes silencing defects at the intergenic spacer sequences of ribosomal genes during interphase and at Y′ repeats in subtelomeric regions during mitosis. We show that the role of Cdc14 in silencing is independent of the RENT deacetylase subunit Sir2. Instead, Cdc14 acts directly on RNA polymerase II by targeting CTD phosphorylation at Ser 2 and Ser 5. We also find that the role of Cdc14 as a CTD phosphatase is conserved in humans. Finally, telomere segregation defects in cdc14 mutants4 correlate with the presence of subtelomeric Y′ elements and can be rescued by transcriptional inhibition of RNA polymerase II.