Secondary structure and distribution of fusogenic LV-peptides in lipid membranes
24-Nov-2007
European Biophysics Journal, 2007, 37, 4, 435 - 45 published on 24.11.2007
European Biophysics Journal, online article
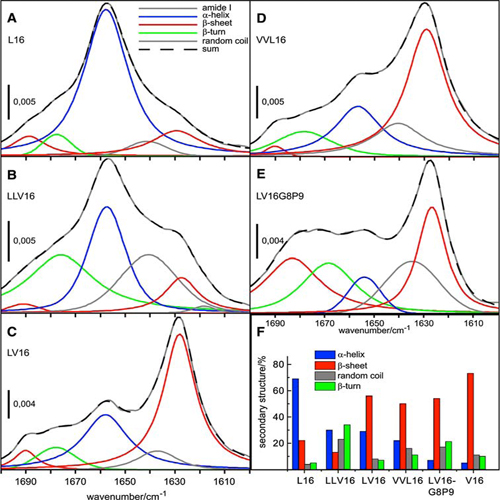
LV-peptides were designed as membrane-spanning low-complexity model structures that mimic fusion protein transmembrane domains. These peptides harbor a hydrophobic core sequence that consists of helix-promoting and helix-destabilizing residues at different ratios. Previously, the fusogenicity of these peptides has been shown to increase with the conformational flexibility of their hydrophobic cores as determined in isotropic solution. Here, we examined the secondary structure, orientation, and distribution of LV-peptides in membranes. Our results reveal that the peptides are homogeneously distributed within the membranes of giant unilamellar liposomes and capable of fusing them. Increasing the valine content of the core up to the level of the β-branched residue content of SNARE TMDs (∼50%) enhances fusogenicity while maintaining a largely α-helical structure in liposomal membranes. A further increase in valine content or introduction of a glycine/proline pair favors β-sheet formation. In planar bilayers, the α-helices adopt oblique angles relative to the bilayer normal and the ratio of α-helix to β-sheet responds more sensitively to valine content. We propose that the fusogenic conformation of LV-peptides is likely to correspond to a membrane-spanning α-helix. β-Sheet formation in membranes may be considered a side-reaction whose extent reflects conformational flexibility of the core.