The histone chaperone sNASP binds a conserved peptide motif within the globular core of histone H3 through its TPR repeats
15-Dec-2015
Nucl. Acids Res., doi: 10.1093/nar/gkv1372
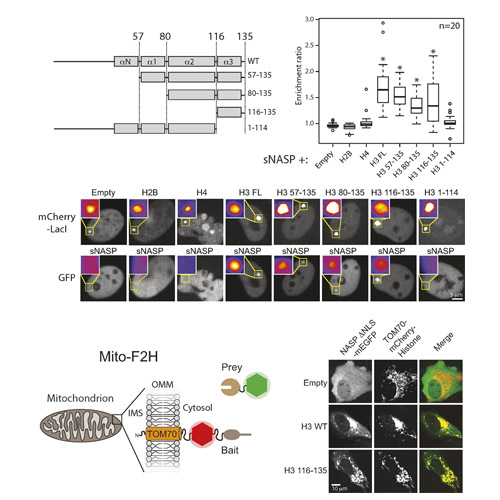
Eukaryotic chromatin is a complex yet dynamic structure, which is regulated in part by the assembly and disassembly of nucleosomes. Key to this process is a group of proteins termed histone chaperones that guide the thermodynamic assembly of nucleosomes by interacting with soluble histones. Here we investigate the interaction between the histone chaperone sNASP and its histone H3 substrate. We find that sNASP binds with nanomolar affinity to a conserved heptapeptide motif in the globular domain of H3, close to the C-terminus. Through functional analysis of sNASP homologues we identified point mutations in surface residues within the TPR domain of sNASP that disrupt H3 peptide interaction, but do not completely disrupt binding to full length H3 in cells, suggesting that sNASP interacts with H3 through additional contacts. Furthermore, chemical shift perturbations from 1H-15N HSQC experiments show that H3 peptide binding maps to the helical groove formed by the stacked TPR motifs of sNASP. Our findings reveal a new mode of interaction between a TPR repeat domain and an evolutionarily conserved peptide motif found in canonical H3 and in all histone H3 variants, including CenpA and have implications for the mechanism of histone chaperoning within the cell.