Structural Analysis of Spiro b-Lactone Proteasome Inhibitors
17-Oct-2008
J. Am. Chem. Soc., 2008, 130 (45), 14981-3 published on 17.10.2008
J. Am. Chem. Soc., online article
J. Am. Chem. Soc., online article
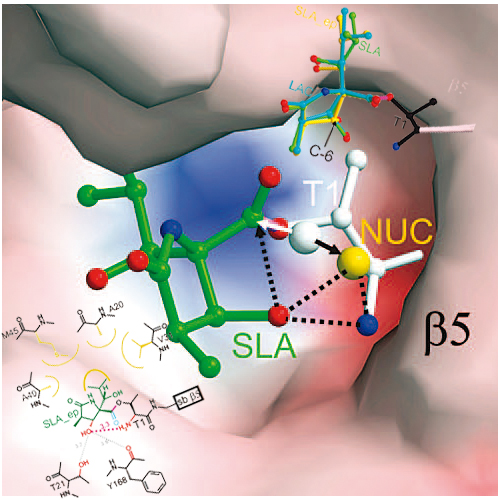
Spiro β-lactone-based proteasome inhibitors were discovered in the context of an asymmetric catalytic total synthesis of the natural product (+)-lactacystin (1). Lactone 4 was found to be a potent inhibitor of the 26S proteasome, while its C-6 epimer (5) displayed weak activity. Crystallographic studies of the two analogues covalently bound to the 20S proteasome permitted characterization of the important stabilizing interactions between each inhibitor and the proteasome’s key catalytic N-terminal threonine residue. This structural data support the hypothesis that the discrepancy in potency between 4 and 5 may be due to differences in the hydrolytic stabilities of the resulting acyl enzyme complexes.